|
Various stimuli have been linked to the activation of NLRP1.[{{Cite journal |last1=Chen |first1=Chen |last2=Xu |first2=Pinglong |date=2022-07-20 |title=Activation and Pharmacological Regulation of Inflammasomes |journal=Biomolecules |language=en |volume=12 |issue=7 |page=1005 |doi=10.3390/biom12071005 |doi-access=free |issn=2218-273X |pmc=9313256 |pmid=35883561}}][{{cite journal |display-authors=6 |vauthors=Bruey JM, Bruey-Sedano N, Luciano F, Zhai D, Balpai R, Xu C, Kress CL, Bailly-Maitre B, Li X, Osterman A, Matsuzawa S, Terskikh AV, Faustin B, Reed JC |date=April 2007 |title=Bcl-2 and Bcl-XL regulate proinflammatory caspase-1 activation by interaction with NALP1 |journal=Cell |volume=129 |issue=1 |pages=45–56 |doi=10.1016/j.cell.2007.01.045 |pmid=17418785 |s2cid=18347164 |doi-access=free}}] |
|
Various stimuli have been linked to the activation of NLRP1.[{{Cite journal |last1=Chen |first1=Chen |last2=Xu |first2=Pinglong |date=2022-07-20 |title=Activation and Pharmacological Regulation of Inflammasomes |journal=Biomolecules |language=en |volume=12 |issue=7 |page=1005 |doi=10.3390/biom12071005 |doi-access=free |issn=2218-273X |pmc=9313256 |pmid=35883561}}][{{cite journal |display-authors=6 |vauthors=Bruey JM, Bruey-Sedano N, Luciano F, Zhai D, Balpai R, Xu C, Kress CL, Bailly-Maitre B, Li X, Osterman A, Matsuzawa S, Terskikh AV, Faustin B, Reed JC |date=April 2007 |title=Bcl-2 and Bcl-XL regulate proinflammatory caspase-1 activation by interaction with NALP1 |journal=Cell |volume=129 |issue=1 |pages=45–56 |doi=10.1016/j.cell.2007.01.045 |pmid=17418785 |s2cid=18347164 |doi-access=free}}] |
|
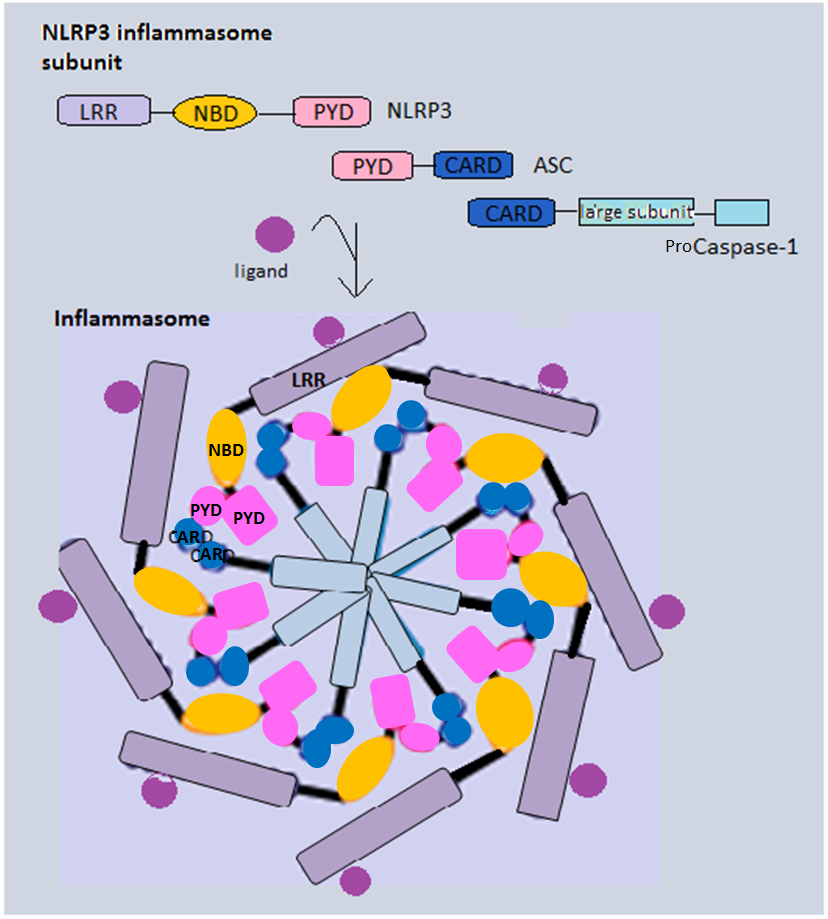
NLRP3 is one of the best-researched inflammasome sensors. The NLRP3 structure consists of PYD along with the NOD and LRR domains. It triggers caspase-1 activation by using PYD to recruit ASC and form a single oligomer in each cell, which includes ten NLRP3 molecules in humans.[{{Cite journal |last1=Fu |first1=Jianing |last2=Wu |first2=Hao |date=2023-04-26 |title=Structural Mechanisms of NLRP3 Inflammasome Assembly and Activation |journal=Annual Review of Immunology |language=en |volume=41 |issue=1 |pages=301–316 |doi=10.1146/annurev-immunol-081022-021207 |issn=0732-0582 |pmc=10159982 |pmid=36750315}}] NLRP3 has been recognized as the largest inflammasome, with a diameter of around 1–2 μm.[{{cite journal |vauthors=Stutz A, Golenbock DT, Latz E |date=December 2009 |title=Inflammasomes: too big to miss |journal=The Journal of Clinical Investigation |volume=119 |issue=12 |pages=3502–3511 |doi=10.1172/JCI40599 |pmc=2786809 |pmid=19955661}}][{{Cite journal |last1=Kelley |first1=Nathan |last2=Jeltema |first2=Devon |last3=Duan |first3=Yanhui |last4=He |first4=Yuan |date=2019-07-06 |title=The NLRP3 Inflammasome: An Overview of Mechanisms of Activation and Regulation |journal=International Journal of Molecular Sciences |language=en |volume=20 |issue=13 |page=3328 |doi=10.3390/ijms20133328 |doi-access=free |issn=1422-0067 |pmc=6651423 |pmid=31284572 |bibcode=2019IJMSc..20.3328K }}][{{Cite journal |last1=Nagar |first1=Abhinit |last2=Bharadwaj |first2=Ravi |last3=Shaikh |first3=Mohammad Omar Faruk |last4=Roy |first4=Abhishek |date=2023-07-26 |title=What are NLRP3-ASC specks? an experimental progress of 22 years of inflammasome research |journal=Frontiers in Immunology |volume=14 |article-number=1188864 |doi=10.3389/fimmu.2023.1188864 |doi-access=free |issn=1664-3224 |pmc=10411722 |pmid=37564644}}] |
|
NLRP3 is one of the best-researched inflammasome sensors. The NLRP3 structure consists of PYD along with the NOD and LRR domains. It triggers caspase-1 activation by using PYD to recruit ASC and form a single oligomer in each cell, which includes ten NLRP3 molecules in humans.[{{Cite journal |last1=Fu |first1=Jianing |last2=Wu |first2=Hao |date=2023-04-26 |title=Structural Mechanisms of NLRP3 Inflammasome Assembly and Activation |journal=Annual Review of Immunology |language=en |volume=41 |issue=1 |pages=301–316 |doi=10.1146/annurev-immunol-081022-021207 |issn=0732-0582 |pmc=10159982 |pmid=36750315}}] NLRP3 has been recognized as the largest inflammasome, with a diameter of around 1–2 μm.[{{cite journal |vauthors=Stutz A, Golenbock DT, Latz E |date=December 2009 |title=Inflammasomes: too big to miss |journal=The Journal of Clinical Investigation |volume=119 |issue=12 |pages=3502–3511 |doi=10.1172/JCI40599 |pmc=2786809 |pmid=19955661}}][{{Cite journal |last1=Kelley |first1=Nathan |last2=Jeltema |first2=Devon |last3=Duan |first3=Yanhui |last4=He |first4=Yuan |date=2019-07-06 |title=The NLRP3 Inflammasome: An Overview of Mechanisms of Activation and Regulation |journal=International Journal of Molecular Sciences |language=en |volume=20 |issue=13 |page=3328 |doi=10.3390/ijms20133328 |doi-access=free |issn=1422-0067 |pmc=6651423 |pmid=31284572 |bibcode=2019IJMSc..20.3328K }}][{{Cite journal |last1=Nagar |first1=Abhinit |last2=Bharadwaj |first2=Ravi |last3=Shaikh |first3=Mohammad Omar Faruk |last4=Roy |first4=Abhishek |date=2023-07-26 |title=What are NLRP3-ASC specks? an experimental progress of 22 years of inflammasome research |journal=Frontiers in Immunology |volume=14 |article-number=1188864 |doi=10.3389/fimmu.2023.1188864 |doi-access=free |issn=1664-3224 |pmc=10411722 |pmid=37564644}}] |
|
NLRP3 oligomerization is activated by a large number of diverse stimuli, such as DAMPs, including crystalline matter (e.g., monosodium urate (MSU) crystals), alum, asbestos, calcium influx, mitochondrial reactive oxygen species (ROS), and extracellular ATP.[{{cite book |title=Inflammasome Signaling and Bacterial Infections |vauthors=Crowley SM, Knodler LA, Vallance BA |date=2016 |publisher=Springer International Publishing |isbn=978-3-319-41170-5 |veditors=Backert S |series=Current Topics in Microbiology and Immunology |volume=397 |pages=43–67 |chapter=Salmonella and the Inflammasome: Battle for Intracellular Dominance |doi=10.1007/978-3-319-41171-2_3 |pmid=27460804}}] The NLRP3 inflammasome is additionally known to assemble in response to PAMPs from pathogens including viruses such as [[Influenza A virus|influenza A]],[{{Cite journal |last1=Kanneganti |first1=Thirumala-Devi |last2=Body-Malapel |first2=Mathilde |last3=Amer |first3=Amal |last4=Park |first4=Jong-Hwan |last5=Whitfield |first5=Joel |last6=Franchi |first6=Luigi |last7=Taraporewala |first7=Zenobia F. |last8=Miller |first8=David |last9=Patton |first9=John T. |last10=Inohara |first10=Naohiro |last11=Núñez |first11=Gabriel |date=December 2006 |title=Critical Role for Cryopyrin/Nalp3 in Activation of Caspase-1 in Response to Viral Infection and Double-stranded RNA |journal=Journal of Biological Chemistry |language=en |volume=281 |issue=48 |pages=36560–36568 |doi=10.1074/jbc.M607594200|doi-access=free |pmid=17008311 }}] bacteria such as ''[[Neisseria gonorrhoeae]]''[30] and bacterial toxins such as [[nigericin]] and [[maitotoxin]],[{{Cite journal |last1=Saïd-Sadier |first1=Najwane |last2=Padilla |first2=Eduardo |last3=Langsley |first3=Gordon |last4=Ojcius |first4=David M. |date=2010-04-02 |editor-last=Unutmaz |editor-first=Derya |title=Aspergillus fumigatus Stimulates the NLRP3 Inflammasome through a Pathway Requiring ROS Production and the Syk Tyrosine Kinase |journal=PLOS ONE |language=en |volume=5 |issue=4 |article-number=e10008 |doi=10.1371/journal.pone.0010008 |doi-access=free |issn=1932-6203 |pmc=2848854 |pmid=20368800|bibcode=2010PLoSO...510008S }}] |
|
NLRP3 oligomerization is activated by a large number of diverse stimuli, such as DAMPs, including crystalline matter (e.g., monosodium urate (MSU) crystals), alum, asbestos, calcium influx, mitochondrial reactive oxygen species (ROS), and extracellular ATP.[{{cite book |title=Inflammasome Signaling and Bacterial Infections |vauthors=Crowley SM, Knodler LA, Vallance BA |date=2016 |publisher=Springer International Publishing |isbn=978-3-319-41170-5 |veditors=Backert S |series=Current Topics in Microbiology and Immunology |volume=397 |pages=43–67 |chapter=Salmonella and the Inflammasome: Battle for Intracellular Dominance |doi=10.1007/978-3-319-41171-2_3 |pmid=27460804}}] The NLRP3 inflammasome is additionally known to assemble in response to PAMPs from pathogens including viruses such as [[Influenza A virus|influenza A]],[{{Cite journal |last1=Kanneganti |first1=Thirumala-Devi |last2=Body-Malapel |first2=Mathilde |last3=Amer |first3=Amal |last4=Park |first4=Jong-Hwan |last5=Whitfield |first5=Joel |last6=Franchi |first6=Luigi |last7=Taraporewala |first7=Zenobia F. |last8=Miller |first8=David |last9=Patton |first9=John T. |last10=Inohara |first10=Naohiro |last11=Núñez |first11=Gabriel |date=December 2006 |title=Critical Role for Cryopyrin/Nalp3 in Activation of Caspase-1 in Response to Viral Infection and Double-stranded RNA |journal=Journal of Biological Chemistry |language=en |volume=281 |issue=48 |pages=36560–36568 |doi=10.1074/jbc.M607594200|doi-access=free |pmid=17008311 }}] bacteria such as ''[[Neisseria gonorrhoeae]]''[30] and bacterial toxins such as [[nigericin]] and [[maitotoxin]],[{{Cite journal |last1=Saïd-Sadier |first1=Najwane |last2=Padilla |first2=Eduardo |last3=Langsley |first3=Gordon |last4=Ojcius |first4=David M. |date=2010-04-02 |editor-last=Unutmaz |editor-first=Derya |title=Aspergillus fumigatus Stimulates the NLRP3 Inflammasome through a Pathway Requiring ROS Production and the Syk Tyrosine Kinase |journal=PLOS ONE |language=en |volume=5 |issue=4 |article-number=e10008 |doi=10.1371/journal.pone.0010008 |doi-access=free |issn=1932-6203 |pmc=2848854 |pmid=20368800|bibcode=2010PLoSO...510008S }}] |
|
Many NLRP3 activators induce cytosolic potassium efflux from cells, and a sufficiently low cytosolic potassium concentration can trigger NLRP3 activation in the absence of other stimuli.[{{Cite journal |last1=Groß |first1=Christina J. |last2=Mishra |first2=Ritu |last3=Schneider |first3=Katharina S. |last4=Médard |first4=Guillaume |last5=Wettmarshausen |first5=Jennifer |last6=Dittlein |first6=Daniela C. |last7=Shi |first7=Hexin |last8=Gorka |first8=Oliver |last9=Koenig |first9=Paul-Albert |last10=Fromm |first10=Stephan |last11=Magnani |first11=Giovanni |last12=Ćiković |first12=Tamara |last13=Hartjes |first13=Lara |last14=Smollich |first14=Joachim |last15=Robertson |first15=Avril A.B. |date=October 2016 |title=K + Efflux-Independent NLRP3 Inflammasome Activation by Small Molecules Targeting Mitochondria |url=https://linkinghub.elsevier.com/retrieve/pii/S107476131630334X |journal=Immunity |language=en |volume=45 |issue=4 |pages=761–773 |doi=10.1016/j.immuni.2016.08.010|pmid=27692612 }}][{{cite journal |vauthors=Jamilloux Y, Sève P, Henry T |date=November 2014 |title=[Inflammasomes in human diseases] |journal=La Revue de Médecine Interne |volume=35 |issue=11 |pages=730–741 |doi=10.1016/j.revmed.2014.04.017 |pmid=24907108}}] This process is thought to be abrogated in atherosclerosis and gout, where these crystals accumulate within the cell. It has also been established that inorganic particles like titanium dioxide, silicon dioxide, and asbestos can activate the inflammasome.[{{cite journal |vauthors=Yazdi AS, Guarda G, Riteau N, Drexler SK, Tardivel A, Couillin I, Tschopp J |date=November 2010 |title=Nanoparticles activate the NLR pyrin domain containing 3 (Nlrp3) inflammasome and cause pulmonary inflammation through release of IL-1α and IL-1β |journal=Proceedings of the National Academy of Sciences of the United States of America |volume=107 |issue=45 |pages=19449–19454 |bibcode=2010PNAS..10719449Y |doi=10.1073/pnas.1008155107 |pmc=2984140 |pmid=20974980 |doi-access=free}}] While the activation of the NLRP3 inflammasome leads to a potent inflammatory response to various pathogens and stress signals,[{{cite journal |display-authors=6 |vauthors=Zielinski MR, Gerashchenko D, Karpova SA, Konanki V, McCarley RW, Sutterwala FS, Strecker RE, Basheer R |date=May 2017 |title=The NLRP3 inflammasome modulates sleep and NREM sleep delta power induced by spontaneous wakefulness, sleep deprivation and lipopolysaccharide |journal=Brain, Behavior, and Immunity |volume=62 |pages=137–150 |doi=10.1016/j.bbi.2017.01.012 |pmc=5373953 |pmid=28109896}}] Additionally, recent studies indicate that NLRP3 inflammasome-mediated neuroinflammation is involved in secondary brain injury following intracerebral hemorrhage.[{{cite journal |display-authors=6 |vauthors=Ren H, Han R, Chen X, Liu X, Wan J, Wang L, Yang X, Wang J |date=September 2020 |title=Potential therapeutic targets for intracerebral hemorrhage-associated inflammation: An update |journal=Journal of Cerebral Blood Flow and Metabolism |volume=40 |issue=9 |pages=1752–1768 |doi=10.1177/0271678X20923551 |pmc=7446569 |pmid=32423330 |s2cid=218689863}}] ''In addition to the well-characterized NLRP3, the NLRP2 inflammasome has also been identified as a significant regulator in the central nervous system. It is notably expressed in astrocytes and has been linked to neuroinflammatory pathways involved in Alzheimer's disease and chronic pain.'' |
|
Many NLRP3 activators induce cytosolic potassium efflux from cells, and a sufficiently low cytosolic potassium concentration can trigger NLRP3 activation in the absence of other stimuli.[{{Cite journal |last1=Groß |first1=Christina J. |last2=Mishra |first2=Ritu |last3=Schneider |first3=Katharina S. |last4=Médard |first4=Guillaume |last5=Wettmarshausen |first5=Jennifer |last6=Dittlein |first6=Daniela C. |last7=Shi |first7=Hexin |last8=Gorka |first8=Oliver |last9=Koenig |first9=Paul-Albert |last10=Fromm |first10=Stephan |last11=Magnani |first11=Giovanni |last12=Ćiković |first12=Tamara |last13=Hartjes |first13=Lara |last14=Smollich |first14=Joachim |last15=Robertson |first15=Avril A.B. |date=October 2016 |title=K + Efflux-Independent NLRP3 Inflammasome Activation by Small Molecules Targeting Mitochondria |url=https://linkinghub.elsevier.com/retrieve/pii/S107476131630334X |journal=Immunity |language=en |volume=45 |issue=4 |pages=761–773 |doi=10.1016/j.immuni.2016.08.010|pmid=27692612 }}][{{cite journal |vauthors=Jamilloux Y, Sève P, Henry T |date=November 2014 |title=[Inflammasomes in human diseases] |journal=La Revue de Médecine Interne |volume=35 |issue=11 |pages=730–741 |doi=10.1016/j.revmed.2014.04.017 |pmid=24907108}}] This process is thought to be abrogated in atherosclerosis and gout, where these crystals accumulate within the cell. It has also been established that inorganic particles like titanium dioxide, silicon dioxide, and asbestos can activate the inflammasome.[{{cite journal |vauthors=Yazdi AS, Guarda G, Riteau N, Drexler SK, Tardivel A, Couillin I, Tschopp J |date=November 2010 |title=Nanoparticles activate the NLR pyrin domain containing 3 (Nlrp3) inflammasome and cause pulmonary inflammation through release of IL-1α and IL-1β |journal=Proceedings of the National Academy of Sciences of the United States of America |volume=107 |issue=45 |pages=19449–19454 |bibcode=2010PNAS..10719449Y |doi=10.1073/pnas.1008155107 |pmc=2984140 |pmid=20974980 |doi-access=free}}] While the activation of the NLRP3 inflammasome leads to a potent inflammatory response to various pathogens and stress signals,[{{cite journal |display-authors=6 |vauthors=Zielinski MR, Gerashchenko D, Karpova SA, Konanki V, McCarley RW, Sutterwala FS, Strecker RE, Basheer R |date=May 2017 |title=The NLRP3 inflammasome modulates sleep and NREM sleep delta power induced by spontaneous wakefulness, sleep deprivation and lipopolysaccharide |journal=Brain, Behavior, and Immunity |volume=62 |pages=137–150 |doi=10.1016/j.bbi.2017.01.012 |pmc=5373953 |pmid=28109896}}] Additionally, recent studies indicate that NLRP3 inflammasome-mediated neuroinflammation is involved in secondary brain injury following intracerebral hemorrhage.[{{cite journal |display-authors=6 |vauthors=Ren H, Han R, Chen X, Liu X, Wan J, Wang L, Yang X, Wang J |date=September 2020 |title=Potential therapeutic targets for intracerebral hemorrhage-associated inflammation: An update |journal=Journal of Cerebral Blood Flow and Metabolism |volume=40 |issue=9 |pages=1752–1768 |doi=10.1177/0271678X20923551 |pmc=7446569 |pmid=32423330 |s2cid=218689863}}] |
|
The NLRP3 inflammasome is also an essential component of several PANoptosomes, including the ZBP1-, RIPK1-, and NLRP12-PANoptosome.[{{Cite journal |last1=Pandeya |first1=Ankit |last2=Kanneganti |first2=Thirumala-Devi |date=January 2024 |title=Therapeutic potential of PANoptosis: innate sensors, inflammasomes, and RIPKs in PANoptosomes |journal=Trends in Molecular Medicine |language=en |volume=30 |issue=1 |pages=74–88 |doi=10.1016/j.molmed.2023.10.001 |pmc=10842719 |pmid=37977994}}] ''I'' |
|
The NLRP3 inflammasome is also an essential component of several PANoptosomes, including the ZBP1-, RIPK1-, and NLRP12-PANoptosome.[{{Cite journal |last1=Pandeya |first1=Ankit |last2=Kanneganti |first2=Thirumala-Devi |date=January 2024 |title=Therapeutic potential of PANoptosis: innate sensors, inflammasomes, and RIPKs in PANoptosomes |journal=Trends in Molecular Medicine |language=en |volume=30 |issue=1 |pages=74–88 |doi=10.1016/j.molmed.2023.10.001 |pmc=10842719 |pmid=37977994}}] |

 Like
0
Like
0
 Dislike
0
Dislike
0
 Love
0
Love
0
 Funny
0
Funny
0
 Angry
0
Angry
0
 Sad
0
Sad
0
 Wow
0
Wow
0